SLS Administration Tool (AT)
Formulary Management
Overview
Codonics award-winning Safe Label System (SLS) is the world’s leading solution for safe, compliant, and fast medication labeling — anywhere medications are prepared! An FDA Class 2 medication safety system, SLS uses barcode technology to improve safety, compliance and efficiency during medication preparation. SLS helps to eliminate vial and ampoule swaps, mislabeling, and syringe swaps in the OR or anywhere medications are prepared.
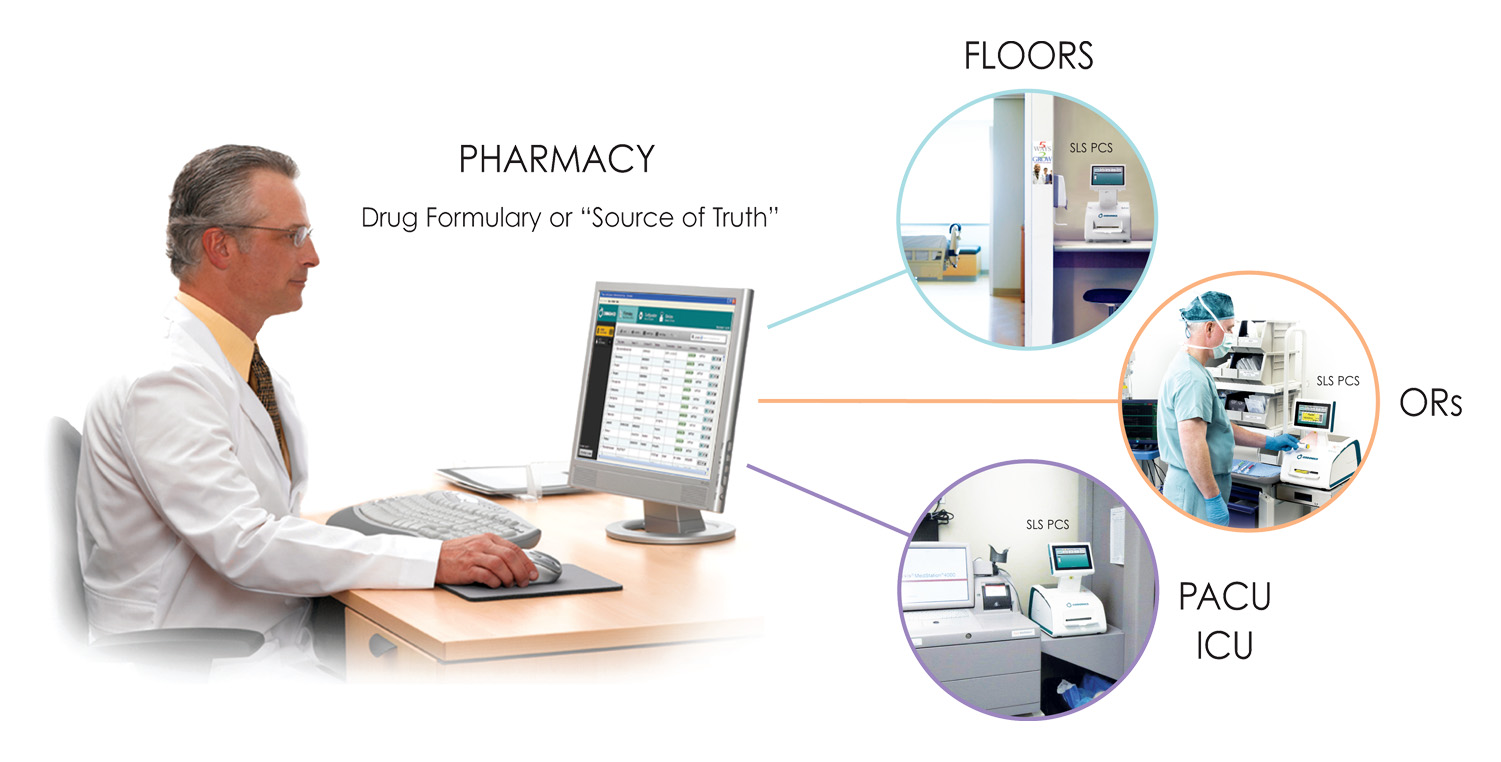
The system has two unique components. The Administration Tool (AT) software is used in the pharmacy while the SLS hardware is deployed to the point of care — like the OR — and brings pharmacy and anesthesia together to better manage medication use to improve patient safety.
SLS Administration Tool
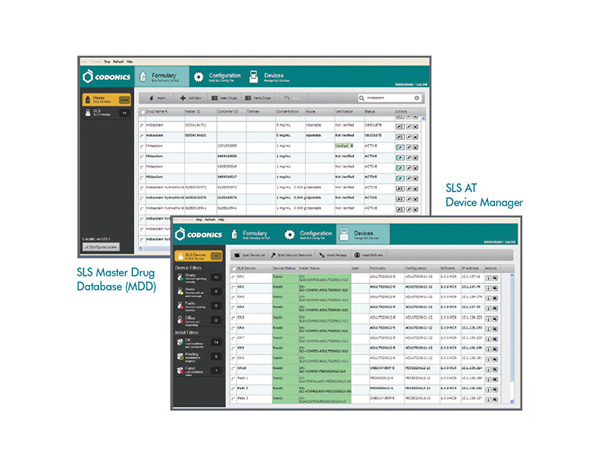
At the heart of the SLS is the Administration Tool (AT), an easy-to-use software application for formulary management in pharmacy. The AT allows pharmacy to easily create and control a drug formulary. As a formulary builder, the AT enables formularies specific to OR, SICU, PICU, ICU and other patient care areas to be enterprise networked for easy access at every SLS system, expanding pharmacy’s scope of influence without having to have a pharmacist present everywhere medications are prepared.
Pharmacy oversight in the OR
Never before has it been easier to empower every medication preparation area with pharmacy-level oversight and compliance. The AT allows pharmacy to easily create and control a drug formulary, including site-approved medications, concentrations and rules for use, expanding pharmacy’s scope of influence to the point of medication preparation.
For the first time, SLS AT makes it practical and easy for pharmacy to control and manage distribution of drugs, drug use rules and drug information throughout a hospital (to literally hundreds of locations with a single click of a button).
Standardization drives safety
As a pharmacist, you have the ultimate responsibility for medication usage everywhere in the hospital, including the operating room. In many facilities, pharmacy struggles with how to get standardization and compliance into this fast-paced environment. Not anymore! In a few clicks, the SLS Administration Tool consolidates your approved drug database and rules – such as whether or not a drug can be diluted and what dilution concentrations are approved. It’s merged with the Joint Commission, ASA and USP accreditation and compliancy requirements to create a site-specific formulary. Each SLS accesses that database every time a drug is scanned, instantly introducing standardization into the OR. That standardization drives safety.
“I think the greatest benefit to pharmacy is that we don’t have to worry about labeling practice in the OR any longer. When Joint Commission comes or any accredited agency comes, we know that we have a system in place and practice that is considered ‘best in show’ on how we standardize practice in our OR environment and ensure that labeling is happening with the appropriate expiration, concentration, the initials of the provider that actually prepared the medication….it helps us sleep at night.”
-Jeff Wagner, Director of Pharmacy, Texas Children’s Hospital
