Reduce Medication Errors
Safe Label System (SLS)
Medication Labeling Solution
Safe, compliant, fast medication labeling — anywhere medications are prepared
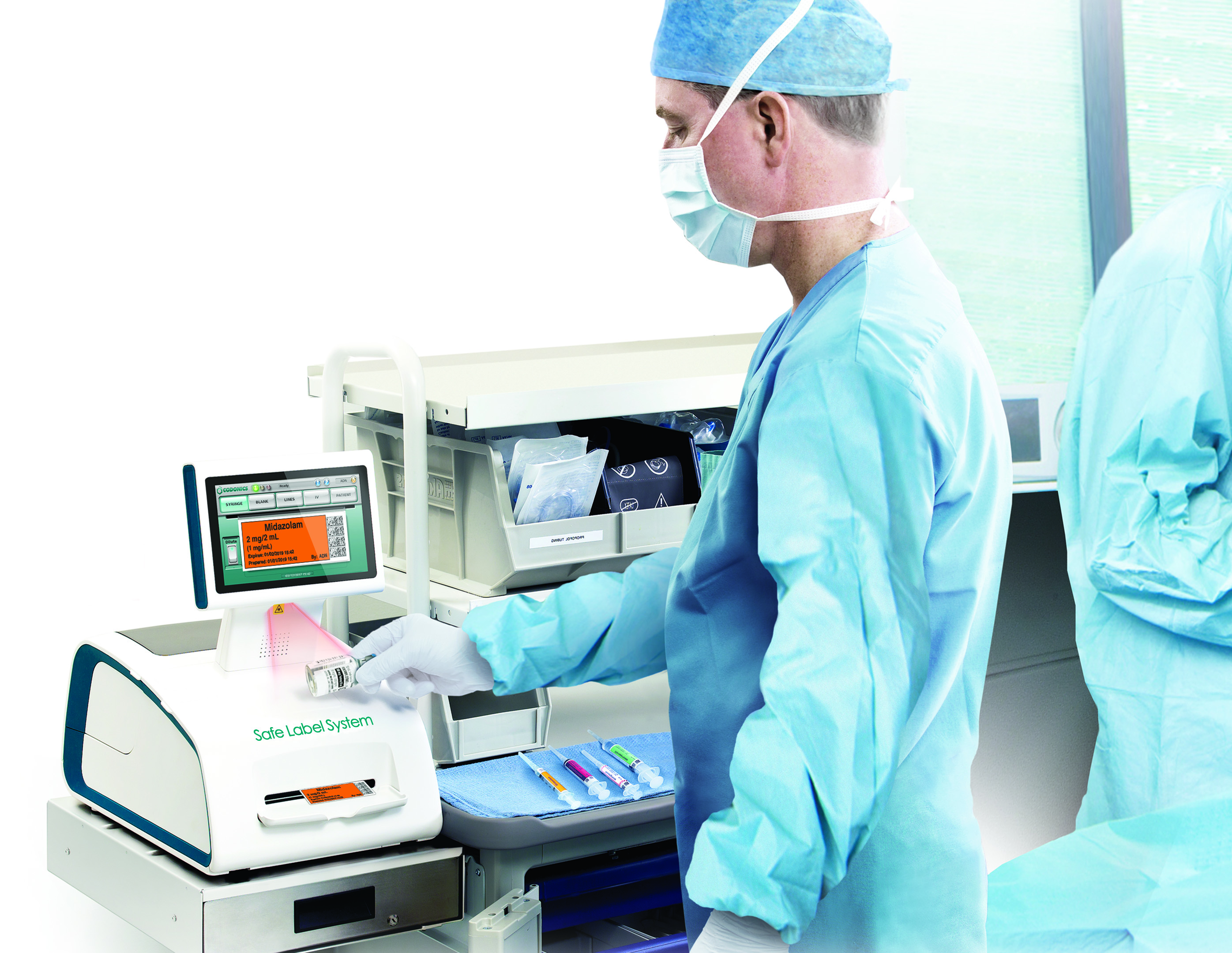
Codonics Safe Label System (SLS) is an award-winning, FDA-approved medical device that improves the safety and accuracy of medication management, integrating worldwide recognized best practices and international standards. A complete solution to enhance patient care, SLS uses barcode technology to read information from a drug container and electronically verify it against the hospital’s pharmacy-approved formulary database at the point of care.
NEW! Safe Label System is now RFID upgradeable. The next generation Safe Label System 600i model “future proofs” your perioperative and procedural areas for RFID. It features all of the capabilities of the SLS 550i, supporting GS1 DataMatrix, 1D barcodes and now RFID. SLS 600i can provide data such as expiration date, lot number and NDC to Epic, Cerner and other AIMS/EHR systems providing track and trace capabilities from the manufacturer to the patient while improving efficiency, accuracy and safety.
Where SLS is used
The system integrates with both manual and smart anesthesia drug carts (ADCs)* in the OR, enabling it to be conveniently situated as part of the anesthesia workflow. SLS is also widely used in the ICU, for patient care on the floor and at the bedside, and in the pharmacy.
Improve patient safety
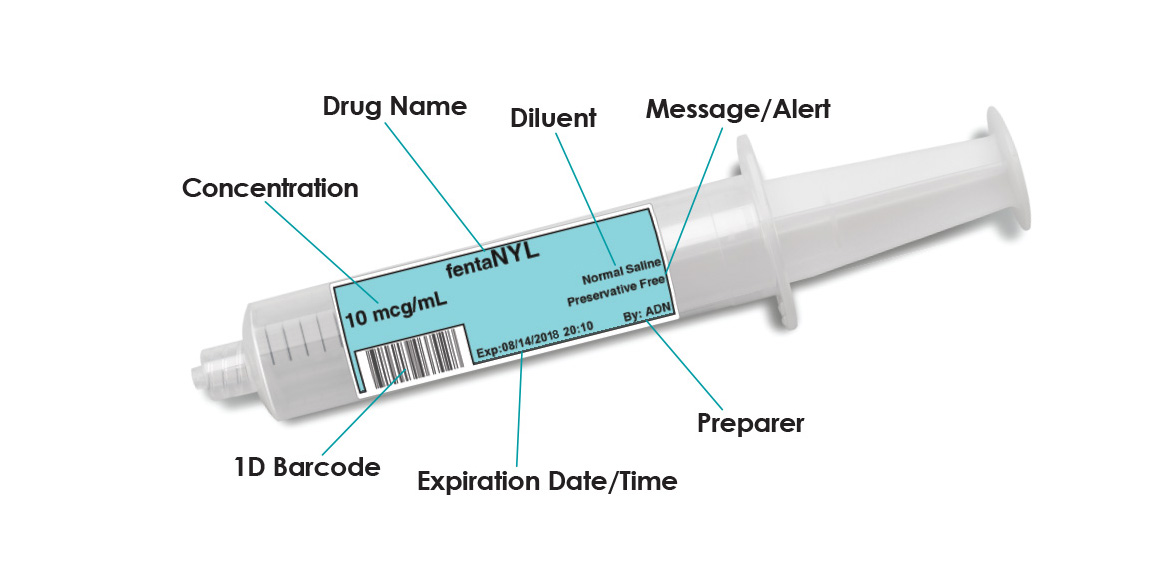
Problems associated with mislabeling and mishandling of medications continue to adversely affect hospitals and surgical facilities. Drug labeling requires time and vigilance, and is prone to human error. For example, in the OR, anesthetists are met with competing demands, coupled with the challenge to handwrite the required drug information in a legible manner on a very small area. SLS uses barcode technology to read information from drug containers and empowers you to label any medication, anywhere in your facility, with a compliant label.
Helps meet ISMP Guidelines
The perioperative and procedural settings are high-risk environments for medication errors1. When medication errors occur, they are often the result of complexity of care, numerous handoffs, fast pace, and medication-related issues2-3. The Institute for Safe Medication Practices (ISMP) has developed Guidelines on medication safety in these environments to help address and reduce the risk of these errors.
The objective of the ISMP Guidelines is to provide hospitals, ambulatory surgery centers and other procedural locations with a comprehensive framework that enables gaps in their medication systems and practices to be preemptively recognized and prioritized making way for implementation of best practices.
Codonics Safe Label System is an effective way to help meet ISMP Guidelines, especially key elements #4 and #10. By following these Guidelines and using Safe Label System to create a standardization for medication safety, healthcare organizations can help improve patient outcomes by reducing the risk of medication errors caused by human error.
Workflow eliminates medication errors and improves compliance
When a drug is scanned on SLS, the clinician is immediately presented with audible and visual confirmation of the drug and concentration in hand, providing an electronic “double-check”, a safeguard that ensures the medication selected is the intended medication. The system then automatically presents a TJC-compliant, easy-to-read and ready-to-apply label that meets ASA guidelines, eliminating the need for handwriting and ensuring safety, accuracy and compliancy.
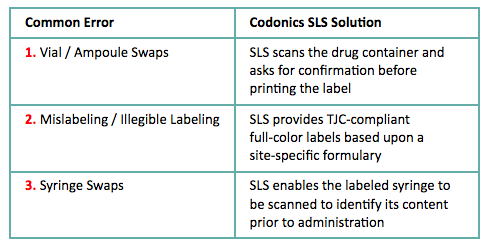
SLS reduces the 3 most common drug errors made in the OR:
Additional benefits
- Includes a 2D barcode on labels enabling electronic documentation of the medication in the patient’s AIMS/EHR
- When integrated with an anesthesia cart, SLS can help to decrease stock-outs, improve inventory control and increase charge capture
- Data analytics provide pharmacy insight into the OR
- When integrated with AIMS, identifies syringes prior to administration, provides warnings for expired syringes, patient allergies or adverse drug interactions
- Prints labels for IV lines, invasive monitor lines, and other OR needs on demand
That’s just the beginning.
From pre-installation assessment to post-installation service and support – plus everything in between– Codonics is there.
Interested in a Safe Label System demo?
Patient Safety is our highest priority
SLS Capabilities
- FDA Class II medical device
- Medication safety system
- Installed base of more than 14,500 in more than 950 sites across 19 countries
- Medication management solution
- Hospital-specific, best practice concentrations pre-set for speed and safety with no manual entry required
- Produces TJC-compliant labels
- Meets Medication Management (MM) 05.01.09 name, strength (concentration) and amount
- Meets NPSG 03.04.01 labeling diluents, volume
- Includes audible/visual identification at preparation and verification at administration, acting as a second set of eyes to ensure safety
- Produces ASA-compliant full-color labels and follows latest Label Enhancements to Reduce Drug Administration Errors: barcoding
- Includes ISO 26825, ASTM 4774-06 and ASTM 4474-11 (including ASTM D4267 and D6398-08; font/characters, contrast and enhanced readability)
- Essential information including the drug’s generic name and concentration barcoded at a location on the label which will not interfere with the label’s legibility as specified in Section 8 of ASTM D6398
- Labels indicate final concentration and diluent if drug is diluted or reconstituted
- Labels include 1D and 2D barcode (for EHR/AIMS integration), providing charge capture and helping to meet 340B requirements
- SLS labels enable drug and concentration to be confirmed prior to administration
- Free-form message fields on printed label (e.g. warnings, hospital address)
- Combination drug templates
- Logon via barcode scan or touchscreen interface
- Requires NDC barcode scan of the medication/container to ensure safety
- Dilute option
- Selectable diluent
- Prints labels for IV, invasive monitor lines, patient labels or customized labels (e.g. pump lines, needle gauges, breast milk, etc.)
- Ability to make multiple labels from a single scan
- Stand-alone operation
- Integrates with/on anesthesia drug carts for convenience during preparation
- Completely eliminates handwriting
- Automates labeling process
- Visual alerts for drug name, concentration, date/time of expiration, recalled drugs and expired drugs
- Audible alerts for drug name, concentration, date/time of expiration, recalled drugs, expired drugs and dilution required
- Helps prevent vial ampoule swaps, mislabeling and syringe swaps
- Integrates with AIMS including Epic, iMDsoft, Plexus, etc.
- Published articles, case studies and white papers (ROI from reduced inventory discrepancies; reduces medication errors while increasing TJC compliance)
- Optional shelf (for integration on ADCs)
- Includes starter kit (ink/labels)
- SLS Advanced “Smart” Scanning (SASS) (supplies that don’t need a label but need to be scanned for inventory control)*
- BCMA integration on the floor and in the OR (Epic, Cerner, Allscripts, McKesson, MEDITECH, GE Healthcare, etc.)
*SLS integration varies with our partners. Ask you sales representative for details.
Solutions by Department

In the Pharmacy
Given today’s demand for safe and effective medication preparation, providing strict oversight is a challenging task for pharmacy, yet essential to ensure medication safety, security and management. Safe Label System (SLS) empowers the hospital with the tools to merge hospital-approved formularies and policies with international labeling standards in high risk procedural areas and anywhere medications are prepared.
In the OR
In the fast-paced enviroment of the OR, ensuring syringes are consistently labeled and in compliance with TJC standards and ASA guidelines can greatly reduce the chance of error. The three most common medication errors which occur in the OR include ampoule and vial swaps, mislabeling or illegible labeling, and syringe swaps.

On the Floor
Despite your dedication to excellent patient care, medication errors are among the most common medical mistakes. To help ensure patient safety, SLS Point of Care Station (PCS) can be placed anywhere medications are prepared – such as patient floors and nurse medication prep stations – providing accurate and compliant medication labels on demand.

In the ICU/PACU
Codonics Safe Label System (SLS) helps reduce the use of costly prefilled syringes by empowering users to prepare medications in the pharmacy. SLS enables medications to be prepared and compliantly labeled in small or large batches and sent up to the floors, and integrates with RIVA robots to improve syringe labeling to meet compliance requirements for use in the perioperative environment.
1. Nanji KC, Patel A, Shaikh S, Seger DL, Bates DW. Evaluation of perioperative medication errors and adverse drug events. Anesthesiology. 2016;124(1):25-34.
2. ISMP launches New Perioperative Medication Safety Guidelines (2022) Institute For Safe Medication Practices. Available at: https://www.ismp.org/news/ismp-launches-new-perioperative-medication-safety-guidelines (Accessed: May 3, 2023).
3. Guidelines for Safe Medication Use in Perioperative and Procedural Settings. Institute For Safe Medication Practices (ISMP) (2022). https://www.ismp.org/system/files/resources/2022-08/ISMP198-Periop-Guidelines-FINAL.pdf